
Authors: Kimberly Wiltrout, MD; Annapurna Poduri, MD, MPH
Boston Children’s Hospital and Harvard Medical School
Reviewed: May 2022
SUMMARY
SYNGAP1-related disorder is a genetic disorder that leads to seizures and developmental problems. The gene that causes the disorder is called SYNGAP1. Certain variants, also called mutations, of this gene can cause a range of neurological symptoms.
The most common symptoms begin in infancy or childhood. They include:
- Developmental delays
- Intellectual disability
- Seizures
- Ataxia (a lack of coordinated movement)
- Autistic features or autism spectrum disorder (ASD)


JUMP TO
Disorder Overview
DESCRIPTION
The SYNGAP1 gene helps make a protein called SynGAP. SynGAP is important for:
- Signaling between nerve cells (neurons)
- Brain development
A disease-causing variant of the SYNGAP1 gene does not allow the body to properly make SynGAP protein. This leads to seizures and other developmental problems.
Children and adults with this disorder may have various combinations of symptoms. However, almost all children with the disorder have developmental delays. Over 80% have epilepsy.
- Developmental Delay. This is typically the first symptom noticed. It is often noted between infancy and age 2.
- Epilepsy. Seizures typically start around age 2. However, they can begin any time from early infancy to later in childhood.
Children with this disorder will have it for their entire lives. Currently, there is no cure for it and no gene-specific therapy. However, there are treatments that can help.
SIGNS AND SYMPTOMS
Symptoms alone cannot lead to a diagnosis of SYNGAP1-related disorder. However, symptoms can lead to genetic testing once they appear. Seizures and other neurologic symptoms are common in this disorder.
Seizures
SYNGAP1-related disorder is linked with several types of seizures.
More common types of seizures include:
Absence seizures.
Absence seizures with eyelid myoclonia.
Atonic seizures.
Tonic-clonic seizures.
Some seizure types are less common with this disorder than with other genetic epilepsies. These include:
Status epilepticus.
Other Symptoms
Other common symptoms of this disorder include:
Behavioral problems.
CAUSES
SYNGAP1-related disorder is caused by a disease-causing variant of the SYNGAP1 gene. The variant typically occurs de novo. De novo means that a new mutation has occurred in the child’s genes. It means that the change was not inherited from either parent.
In rare cases, the SYNGAP1 variant that causes this disorder is inherited from a parent who does not experience the disorder. When this happens, it is due to parental mosaicism. Parental mosaicism occurs when a parent has a small number of cells with the genetic change in low levels. The genetic change may not even be detectable in the parent.
SYNGAP1-related disorder can be caused by several variants of the SYNGAP1 gene. However, even children with the same variant may have different symptoms.
LABORATORY INVESTIGATIONS
The defining feature of this disorder is a genetic difference in the SYNGAP1 gene. Therefore, genetic testing is needed to make a diagnosis. The types of genetic tests that can be used are:
- Exome sequencing. Detects a variant in the sequence of a person’s genes by looking at all known genes. This is useful when doctors are not sure which genes are affected.
- Gene panels. Detects a variant in the sequence of a particular set of selected genes. Note that not all panels have SYNGAP1 on them.
- Chromosome microarray (CMA). Detects if a copy of a gene is missing.
Other tests that are used in patients with this disorder include:
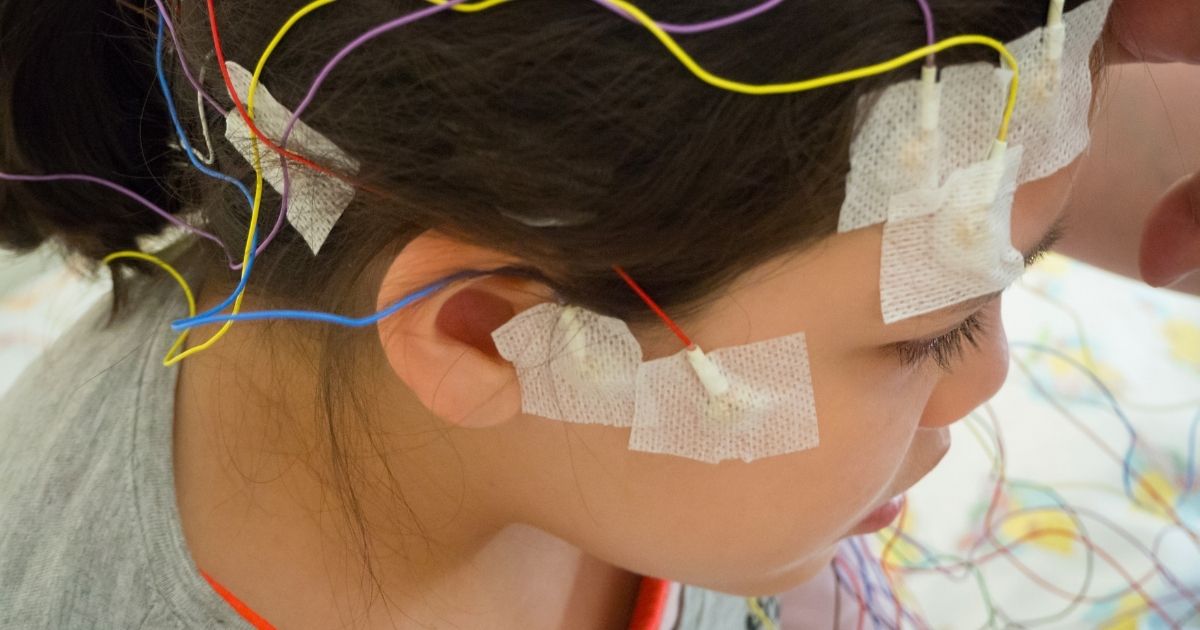
- Electroencephalograms (EEGs). Used to identify the types of seizures that the individual is having.
- Brain magnetic resonance imaging (MRI). Looks for changes in brain structure. The MRI is typically normal in this disorder.
- Blood tests. May be used to monitor medication levels or side effects.
- Neuropsychological testing. Used to test for ASD and intellectual disabilities.

TREATMENT AND THERAPIES
Currently, there is no cure for SYNGAP1–related disorder. There is no specific treatment for the SYNGAP1 gene. However, there are treatments for the symptoms of the disorder. These include:
Antiseizure medication.
The ketogenic diet.
This diet may be used to reduce seizures if they do not improve with medication. The diet is high in fats and low in carbohydrates. Close monitoring is needed for side effects. A special medical team needs to start and monitor patients on this diet. Possible side effects include:
- Vitamin and mineral deficiencies
- Kidney stones
- Weight loss
Surgery.
Therapies.
Therapies help children improve their skills and reach milestones. Typical therapies for children with this disorder include:
- Physical therapy
- Occupational therapy
- Speech therapy
- Applied behavioral analysis (ABA)
Other therapies that may help include:
- Horse therapy
- Aqua therapy
- Music therapy
Research for new treatment, including genetic treatment, is ongoing around the world.


OUTLOOK
There is currently no cure for SYNGAP1-related disorder.
Individuals may experience a range of developmental delays. Most children will be diagnosed with moderate to severe intellectual disability.
Over 80% of those with the disorder have seizures. The seizures can be difficult to control.
Most adults with this disorder are unable to live independently.
RELATED DISORDERS
Someone with SYNGAP1-related disorder may have the types of seizure that resemble a particular epilepsy syndrome. These syndromes include:
- Epilepsy with myoclonic-atonic seizures, also called Doose syndrome
- Epilepsy with eyelid myoclonia, also called Jeavons syndrome
Resources
SynGAP Research Fund (SRF) is a public charity registered in the US, UK & EU. This global group of families is committed to accelerating the science to cure SYNGAP1. The SRF mission is to support the research and development of treatments, therapies, and support systems for SYNGAP1 patients worldwide. You can join the SYNGAP1 Ciitizen Digital Natural History Study to share data for research. Stay up-to-date on the latest information on the SRF Blog page, listen to the podcasts, and follow SRF on all social platforms with the handle @cureSYNGAP1. In addition, SRF hosts SYNGAP1 Global Community, a private Facebook group.
SYNGAP1 Foundation is a 501(c)3 non-profit, patient advocacy organization dedicated to improving the quality of life for patients and families affected by SYNGAP1. The Foundation’s priorities include raising public awareness, patient and family advocacy initiatives, educational programs, scientific research conferences and financial support for research. In addition, the Syngap1 Foundation maintains the largest SYNGAP1 (MRD5) Online Natural History Data Registry in coordination with NORD and with grant funding provided by the FDA. The registry helps to provide the data needed to accelerate SYNGAP1 research. The SYNGAP1 Foundation also hosts a private Facebook group, SYNGAP1 Foundation International Family Connect ~ Find Your Local Tribe.
Simons Searchlight is an ambitious research program that is creating an ever-growing database and resource network. They aim to further the understanding of over 150 genes that cause rare genetic neurodevelopmental disorders, including SYNGAP1. Families share valuable information and experiences that leading geneticists and scientists around the world can use to improve the lives of people living with rare genetic neurodevelopmental disorders. These researchers can also invite families to participate in their research studies in the future. People with genetic diagnoses, their families, and scientists play equal parts in this journey. The study is international, and families can participate in several languages.
Simons Searchlight hosts private Facebook communities for each genetic condition. Connect with the SYNGAP1: Simons Searchlight Community.


Child Neurology Foundation (CNF) solicits resources from the community to be included on this webpage through an application process. CNF reserves the right to remove entities at any time if information is deemed inappropriate or inconsistent with the mission, vision, and values of CNF.
Research
ClinicalTrials.gov for SYNGAP1 (birth to 17 years).
These are clinical trials that are recruiting or will be recruiting. Updates are made daily, so you are encouraged to check back frequently.
ClinicalTrials.gov is a database of privately and publicly funded clinical studies conducted around the world. This is a resource provided by the U.S. National Library of Medicine (NLM), which is an institute within the National Institutes of Health (NIH). Listing a study does not mean it has been evaluated by the U.S. Federal Government. Please read the NLM disclaimer for details.
Before participating in a study, you are encouraged to talk to your health care provider and learn about the risks and potential benefits.
Family Stories
Meet some of the children impacted by SYNGAP1 on the SynGAP Research Fund’s Warriors page. You can also watch inspiring videos of patients on the SRF’s SYNGAP1 Movies page.
SYNGAP1 children and families have many beautiful faces and stories to share. Meet some of these amazing children and families on the SYNGAP1 Foundation website.
The information in the CNF Child Neurology Disorder Directory is not intended to provide diagnosis, treatment, or medical advice and should not be considered a substitute for advice from a healthcare professional. Content provided is for informational purposes only. CNF is not responsible for actions taken based on the information included on this webpage. Please consult with a physician or other healthcare professional regarding any medical or health related diagnosis or treatment options.
References
Agarwal M, Johnston MV, Stafstrom CE. SYNGAP1 mutations: Clinical, genetic, and pathophysiological features. Int J Dev Neurosci. 2019 Nov;78:65-76. https://doi.org/10.1016/j.ijdevneu.2019.08.003. Epub 2019 Aug 24. PMID: 31454529.
Holder JL Jr, Hamdan FF, Michaud JL. SYNGAP1-Related Intellectual Disability. 2019 Feb 21. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Gripp KW, Mirzaa GM, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2022. PMID: 30789692.
Mignot C, von Stülpnagel C, Nava C, Ville D, Sanlaville D, Lesca G, Rastetter A, Gachet B, Marie Y, Korenke GC, Borggraefe I, Hoffmann-Zacharska D, Szczepanik E, Rudzka-Dybała M, Yiş U, Çağlayan H, Isapof A, Marey I, Panagiotakaki E, Korff C, Rossier E, Riess A, Beck-Woedl S, Rauch A, Zweier C, Hoyer J, Reis A, Mironov M, Bobylova M, Mukhin K, Hernandez-Hernandez L, Maher B, Sisodiya S, Kuhn M, Glaeser D, Weckhuysen S, Myers CT, Mefford HC, Hörtnagel K, Biskup S; EuroEPINOMICS-RES MAE working group, Lemke JR, Héron D, Kluger G, Depienne C. Genetic and neurodevelopmental spectrum of SYNGAP1-associated intellectual disability and epilepsy. J Med Genet. 2016 Aug;53(8):511-22. https://doi.org/10.1136/jmedgenet-2015-103451. Epub 2016 Mar 17. Erratum in: J Med Genet. 2016 Oct;53(10):720. PMID: 26989088.
SYNGAP1-related epilepsy [Internet] 2021 Nov [cited 2022 Feb 28]. Available from: https://www.epilepsy.com/learn/epilepsy-due-specific-causes/genetic-causes-epilepsy/specific-genetic-epilepsies/syngap1-related-epilepsy/
SYNGAP1-related NSID [Internet]. (National Organization for Rare Disorders). 2019 [cited 2022 March 9]. Available from: https://rarediseases.org/rare-diseases/syngap1-related-nsid/
Vlaskamp DRM, Shaw BJ, Burgess R, Mei D, Montomoli M, Xie H, Myers CT, Bennett MF, XiangWei W, Williams D, Maas SM, Brooks AS, Mancini GMS, van de Laar IMBH, van Hagen JM, Ware TL, Webster RI, Malone S, Berkovic SF, Kalnins RM, Sicca F, Korenke GC, van Ravenswaaij-Arts CMA, Hildebrand MS, Mefford HC, Jiang Y, Guerrini R, Scheffer IE. SYNGAP1 encephalopathy: A distinctive generalized developmental and epileptic encephalopathy. Neurology. 2019 Jan 8;92(2):e96-e107. https://doi.org/10.1212/WNL.0000000000006729. Epub 2018 Dec 12. Erratum in: Neurology. 2019 Nov 12;93(20):908. PMID: 30541864; PMCID: PMC6340340.
